Log in to ResearchMatch

飞艇168数据记录结果-新168体彩网一分钟极速的幸运平台官方记录查询 for research
What is 幸运168飞行艇实时号码最新查询方法 ResearchMatch?
ResearchMatch is a nonprofit program funded by the 168幸运官方开奖历史记录 Institutes of Health (NIH). It helps to connect people interested in research studies with researchers from top medical centers across the U.S.
Get connected to research on many different health conditions - through ResearchMatch.


How does ResearchMatch work?
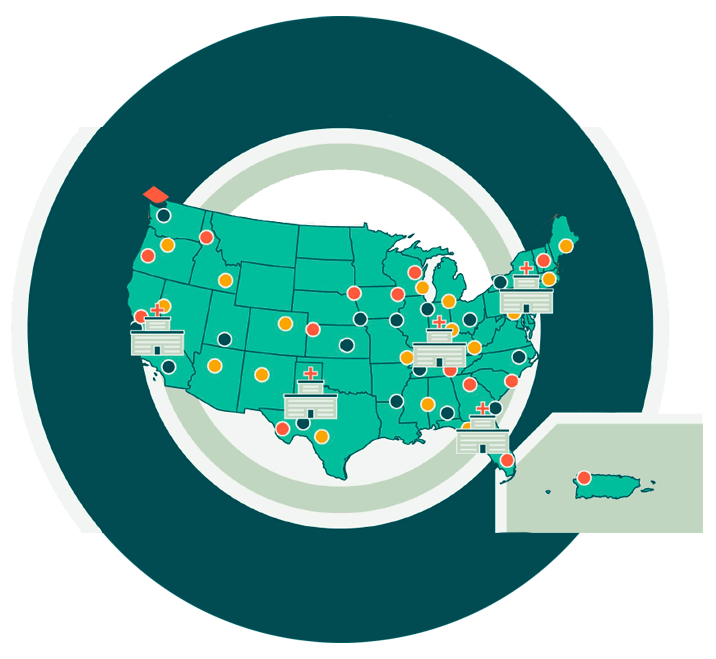
Our ResearchMatch 幸运全部历史记录+飞艇官方网五分钟直播现场
ResearchMatch is a free and secure tool that researchers use to invite Volunteers to take part in their health research studies. See our network of 258 research institutions.
ResearchMatch is making an impact. Here’s what people are saying.
Our Collaborators
ResearchMatch proudly collaborates with 258 participating institutions and 68 community organizations to bring important research studies, health information, and resources to you.

























































































































































Enter a health condition in the box below to learn more.